|
Bakterial biofilms & nanobakterier
Looking for Chinks in the Armor of Bacterial Biofilms. Don Monroe Published: November 13, 2007 Copyright: © 2007 Don Monroe. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Don
Monroe is a freelance science writer living in Berkeley Heights, New Jersey,
United States of America.
Citat från artikeln: "But biofilms are also prime suspects in a long list of other “itises,” including endocarditis, prostatitis, and conjunctivitis."
Biofilms and chronic infections JAMA. 2008 Junc11;299(22):2682-4 Randall D. Wolcott, MD; Garth D. Ehrlich, PhD The prevailing paradigm ofinfectious disease is based on the work of Koch and colleagues, who more than 150 years ago isolated individual strains of bacteria and developed the pure culture-method that is still used today. That work enlightened medicine by firmly establishing the germ-theory of transmissible diseases and demonstrated that diseases like dysentery, tuberculosis and anthrax are caused by microbiological agents.1 Hence, the field of microbiology developed around Koch¢s methods with clinical microbiologists working overwhelmingly with pure log-phase cultures in nutrient-rich media because this approach provided such a powerful tool for the study of acute epidemic bacterial diseases. However, this approach that examines only planktonic bacteria (free-floating, single cell phenotype) may have limited development of a more thorough understanding of microbial processes. In most natural environments and in chronic bacterial infections, the planktonic phenotype generally exists only transiently and usually as a minor population. Emerging evidence describes bacterial populations as predominantly poly-microbial, sessile, community-based aggregations embedded in a self-secreted matrix that provides numerous advantages for persistence in the face of environmental and host-challenges.Therefore, biofilms and the existence of a complex bacterial life-cycle provide a new perspective through which to view infectious diseases. Much of the support for this perspective has come about through the application of new detection- andvisualization-methods that have provided evidence for the theory that chronic infections are fundamentally different than acute infections and that different interventional approaches are necessary to treat these biofilm-infections more efficiently. What Is a Biofilm? A biofilm is a thin layer of micro-organisms that adhere to the surface of an organic or inorganic structure, together with their secreted polymers. Biofilms are the predominant phenotype of nearly all bacteria in their natural habitat, whether pathogenic or environmental. The biofilm provides a bulwark against environmental stressors and can include organisms from multiple kingdoms as in the case of mixed bacterial-fungal biofilms. Thirty years ago, Costerton et al.2 was the first to examine the attributes of biofilms, examining the extracellular polymeric substances (EPS) that holds these community bacteria together. He and other researchers have since implicated biofilms in diverse a-biotic and biotic systems, including oil-pipelines, hot-tubs, teeth and mucosa. Thus biofilms, with their community-defenses, are a ubiquitous feature of bacteria in nature and in some bacterial infections. The resident bacterial community in a biofilm has added defenses and multiple mechanisms for survival, such as defenses against phagocytosis, UV-radiation, viral attack, shear stress and dehydration, as well as against biocides, antibiotics and host-immunity. Biofilms have demonstrated the ability to persist in 100 to 1000 times the concentrations of antibiotics and biocides that can inhibit planktonic cells.3 Similarly, granulocytes, macrophages and other phagocytes are unable to engulf a biofilm as they would individual planktonic cells. The genotypic and phenotypic diversity of the biofilm allow adaptation to overcome multiple stresses and to survive most sequential therapies. The hallmark of biofilms is genetic and phenotypic diversity, which enhances the robustness of the bacterial community.4 An increasing body of evidence suggests that laboratory-cultivated bacteria share few characteristics with infectious biofilms. When bacteria naturally transition from individual planktonic cells to a community of biofilm-tissue, the transcriptional expression of the bacteria radically changes. Although this phenotypic transition is occurring, the bacteria are excreting protective polymers and incorporating environmentalmolecules that bind the bacterial community to a surface and to each other. The biofilm bacterial community uses secreted pheromones (e.g. quorum-sensing molecules) and other molecules for cell-cell signaling, even between species. These coordinated activities render the biofilm a formidable opponent for the host. Biofilms as a Novel Focus. There are 3 significant limitations of continuing to operate within the planktonic paradigm. - First, because planktonic cells have fewer defenses than biofilm-communities, a treatment such as an antibiotic might provide excellent in vitro test-results, when tested on planktonic cells only, but poor in vivo results, in which the biofilm-phenotype may predominate. The same strain of bacteria can be hundreds or thousands of times more resistant to antibiotics if part of a biofilm community.3,5 This planktonic bias undoubtedly accounts for at least some of the discrepancy that can occur between in vitro test results and in vivo response to antibiotic therapy.6 - Second, current culture-methods do not identify the majority of bacteria now known to be in host-infections.7 Researchers have developed molecular, genomic, metagenomic, transcriptomic and proteomic methods because they determined that only approximately 1% of the cells they observed in ecosystems actually produced colonies by conventional culture-methods.8 The conclusion of these DNA-based and RNA-based studies is that culture-methods detect only a small minority of organisms actually present in natural and pathogenic bacterial communities. Therefore, treatment based on conventional cultures may address only 1 or 2 bacterial species in a complex biofilm-community that may include dozens of other species of bacteria, or even of fungi. - Third, planktonic techniques such as culturing may lead to an inaccurate or incomplete diagnosis because cultures do not detect biofilm-cells that are viable but not culturable. Diseases that yield only intermittent positive cultures (e.g. otitis media, prostatitis) have been suggested to be ¡sterile inflammations¢ or viral infections.9 An example of a serious result from reliance on standard cultures is that revisions of the Sulzer acetabular cup continue to be treated as ¡sterile loosenings¢, although this process probably represents a biofilm-infection that does not yield positive cultures but can be detected by molecular methods.10 New Perspectives, Options for Detection and Treatments Changing
the perspective about chronic infectious disease to include biofilm enables 2
important insights. First, it opens new methods for detection and treatment.
Second, it provides a global reconceptualization of many chronic infectious
diseases as resulting from a biofilm, allowing biofilm-principles to be shared
across disciplines. Recent studies have investigated new methods for detecting
the components of a biofilm. Several investigations have used modern molecular
methods, such as denaturing gradient gel electrophoresis and denaturing
high-performance liquid chromatography, along with imaging techniques including
fluorescent in situ hybridization. Also, molecular methods such as polymerase
chain reaction (PCR) and pyro-sequencing in conjunction with conventional
culture-methods have been used to determine the bacterial species composition of
chronic infections.7 Performing molecular tests as part of routine bacterial
analysis is becoming a real option for clinical laboratories. These tests could
include methods such as PCR, reverse transcriptase PCR, micro-arrays,
antigen-testing and rapid sequencing. Only a few of these methods are being used
to test for certain pathogens but culture-free identification of all pathogens
and their corresponding resistance-markers may soon become routine.11 A biofilm
focus also provides new strategies for treatment of chronic infections.
Biofilm-based treatments might Chronic Bacterial Disease as Biofilm Disease Another main benefit the biofilm-model allows is the re-conceptualization of multiple chronic diseases as biofilm-diseases. Biofilm-disease has been viewed as various diseases that affect a variety of tissues and structures, including ear, nose, throat, mouth, eye, lung, heart, kidney, gall-bladder, pancreas, nervous system, skin, bone, as well as virtually every implanted medical device. The Centres for Disease Control and Prevention estimates that more than 65% of infections are caused by bacteria growing in biofilms12 and Lewis13 suggests that the proportion is 80% - by far the majority of infections are estimated to be caused by biofilms. Thus, when biofilm-infections are combined into a single disease category, the prevalence of the disease is significant and the mortality associated with biofilm disease is substantial. Additionally, the disease-processes and management-strategies of biofilms are related. For example, the biofilm-diseases of cystic fibrosis, pneumonia and an infected knee replacement prosthesis are different, but strategies used to manage the biofilm in the lung will be similar to the strategies used for the biofilm on an infected knee-implant. Typical and Familiar Biofilms The typical biofilm-disease manifests with common features. The initial infection is subtle and usually not immediately life-threatening, and antibiotics usually are prescribed. Subsequent exacerbations occur and are again usually treated with antibiotics and adjunct therapies such as corticosteroids. However, the infection worsens when treatment is withdrawn. If the patient¢s clinical status worsens or if the disease progresses to the point that function of the affected tissue or device is sufficiently impaired, a decision often is made to remove the infected tissue or component by surgery. The goal of the physician when addressing the disease is to manage the symptoms and signs, and to suppress exacerbations with the understanding that disease eradication is unlikely without surgical intervention. Perhaps the most commonly encountered biofilm-disease is dental plaque, a condition that can be managed effectively by dental professionals. Periodontal disease and tooth-loss have greatly diminished over the last century due to the multiple concurrent strategies espoused by dentists. The suppression of biofilm in the oral cavity begins with frequent removal of the biofilm by daily brushing and flossing, coupled with periodic dental visits for plaque removal. If the bacterial biofilm load is too great and disease progresses, the frequency of biofilm-management is increased in an attempt to overcome the disease. Dentistry has successfully confronted biofilm-disease and similar approaches may be used as a model for medicine to manage chronic infections. Conclusions Treating chronic infectious diseases cannot be limited to infectious disease specialists any more than the management of diabetes can be limited to endocrinologists. By shifting away from the planktonic model of microbiology to the biofilm-model, new methods for detection and treatment become available. Because of molecular methods, science now has the ability to detect biofilms and understand the implications of interspecies chaos that contribute to infections. With these new scientific approaches along with coordination of clinical and laboratory efforts, education and research, it is possible to imagine overcoming much of biofilm-disease.
Background: Chronic
Prostatitis: Kidney
stones & Astronauts: Polycystic
Kidney Disease (PKD): About
Nanobac Pharmaceuticals: Contact:
Nanobac Test Kit för nanobakterier Nanobac Introduces New Improved ELISA Kits for Detection of Calcifying Nano-Particles, also known as Nanobacteria 0/26/2005 8:00:00 AM EST Nanobac Life Sciences, Inc. (OTCBB: NNBP) ("Nanobac" or "the Company") announced today the introduction of two new versions of its NANOCAPTURE(TM) and NANO-SERO(TM) ELISA test kits for the detection of Calcifying Nano-Particles (CNPs), also known as nanobacteria, in serum or plasma. The new kits offer improved sensitivity, lower background and improved stability over the previous versions. Nanobac has added new standards to improve precision at the low end and freeze dried the critical components for improved stability. "Our demand for CNP diagnostic products is increasing as CNP's or nanobacteria are implicated in more and more chronic diseases," stated Grant Carlson, Nanobac's President and Chief Operating Officer. "Nanobac has worked hard to develop robust, sensitive and reproducible ELISA test kits for the CNP/nanobacteria research community. These kits are CE marked and ready for immediate shipment throughout the European Union. In the United States the kits will be sold For Research Use Only (RUO)." The NANOCAPTURE ELISA is a two-site immunoenzymatic assay for the quantitative measurement of CNP/nanobacterial antigen in serum or plasma. The NANO-SERO ELISA is also a two site immunoenzymatic assay for the quantitative measurement of anti-CNP antibodies in serum or plasma. Nanobac will feature the NANOCAPTURE and NANO-SERO ELISA test kits at the world's #1 medical trade show, MEDICA, in Dusseldorf, Germany, November 16-19, 2005. Last year the show attracted 136,000 visitors from around the world. Nanobac intends to meet with potential customers and establish its global network of in vitro diagnostic distributors. CONTACT:
Allmänt om biofilms vid kronisk prostatit Domingue GJ Sr, Hellstrom WJ. Department of Urology, Tulane University School of Medicine, New Orleans, Louisiana 70112, USA. domingue@mailhost.tcs.tulane.edu The laboratory diagnosis of acute bacterial prostatitis is straightforward and easily accomplished in clinical laboratories. Chronic bacterial prostatitis, and especially chronic idiopathic prostatitis (most often referred to as abacterial prostatitis), presents a real challenge to the clinician and clinical microbiologist. Clinically, the diagnosis of chronic idiopathic prostatitis is differentiated from that of acute prostatitis by a lack of prostatic inflammation and no "significant" (controversial) leukocytes or bacteria in the expressed prostatic secretions. Despite these diagnostic criteria, the etiology of chronic idiopathic prostatitis is unknown. While this review covers the entire spectrum of microbially caused acute prostatitis (including common and uncommon bacteria, viruses, fungi, and parasites) and microbially associated chronic prostatitis, a special focus has been given to chronic idiopathic prostatitis. The idiopathic syndrome is commonly diagnosed in men but is poorly treated. Recent data convincingly suggests a possible bacterial etiology for the condition. Provocative molecular studies have been published reporting the presence of 16S rRNA bacterial sequences in prostate biopsy tissue that is negative for ordinary bacteria by routine culture in men with chronic idiopathic prostatitis. Additionally, special culture methods have indicated that difficult-to-culture coryneforms and coagulase-negative staphylococci are present in expressed prostatic secretions found to be negative by routine culture techniques. Treatment failures are not uncommon in chronic prostatitis. Literature reports suggest that antimicrobial treatment failures in chronic idiopathic prostatitis caused by organisms producing extracellular slime might result from the virulent properties of coagulase-negative staphylococci or other bacteria. While it is difficult to definitively extrapolate from animal models, antibiotic pharmokinetic studies with a murine model have suggested that treatment failures in chronic prostatitis are probably a result of the local microenvironment surrounding the persistent focal and well-protected small bacterial biofilms buried within the prostate gland. These conclusions support the molecular and culture data implicating bacteria as a cause of chronic idiopathic prostatitis. Se även: http://prostatitis.org/biofilms.html där the the Prostatitis Foundation gör en sammanfattning.
J Urol. 2005 Feb;173(2):474-7. Shoskes DA, Thomas KD, Gomez E. From the Department of Kidney Transplantation, Cleveland Clinic Florida, Weston, Florida. PURPOSE: Category III chronic prostatitis/chronic pelvic pain syndrome (CPPS) is a common debilitating condition of unclear etiology. Patients often have prostatic calcifications but a link to symptoms is controversial. Nanobacteria are implicated in stone formation in the urinary tract and, therefore, therapy to eliminate nanobacteria and the stones that they produce might have an impact on CPPS symptoms. MATERIALS AND METHODS: A total of 16 men with recalcitrant CPPS refractory to multiple prior therapies were treated with comET (Nanobac Life Sciences, Tampa, Florida), which consists of 500 mg tetracycline, a proprietary nutraceutical and an ethylenediaminetetraacetic acid suppository daily. The National Institute of Health Chronic Prostatitis Symptom Index (NIH-CPSI), transrectal ultrasound, and blood and urine tests for nanobacterial antigen were performed at the start and conclusion of 3 months of therapy. One patient was lost to followup. RESULTS: Mean NIH-CPSI total score +/- SD decreased from 25.7 +/- 1.6 to 13.7 +/- 2.0 (p <0.0001). Significant improvement was seen in each subscore domain. A total of 12 patients (80%) had at least 25% improvement on NIH-CPSI and 8 (53%) had at least 50% improvement. Nanobacterial antigen or antibody was found in 60% of serum and 40% of urine samples. In 10 patients who underwent transrectal ultrasound after therapy prostatic stones were decreased in size or resolved in 50%. CONCLUSIONS: Therapy designed to eliminate nanobacteria resulted in significant improvement in the symptoms of recalcitrant CPPS in the majority of men, whether due to the treatment of stone producing nanobacteria or through some other mechanism. Prospective placebo controlled trials are warranted. Ovanstående finns i helhet på Journal of Urology http://www.geocities.com/cp_cpps/shoskes-2005-J-Urology.pdf eller http://www.prostatitis.org/pdf/shoskesurology.pdf
Scientists 5/3/2006 11:50:00 AM EST BIOWIRE Nanobac Pharmaceuticals, Inc. (OTCBB:NNBP) ("Nanobac" or "the Company") today announced that Inflammatory nanoparticles that produce calcified deposits like those found in arthritic joints have been cultured from the synovial fluid of rheumatoid arthritis and osteoarthritis patients by Japanese researchers, as reported in the Journal of Proteome Research. "This is the first report to indicate that human synovial fluids contain Nanobacteria-like particles," the study notes. The study, Nanobacteria-Like Particles in Human Arthritic Synovial Fluids, by T. Tsurumoto, T. Matsumoto, A. Yonekura, and H. Shindo, Department of Orthopaedics, Graduate School of Biomedical Sciences, Nagasaki University, supports the existence and pathogenic role of nanobacteria-like particles found in human diseases such as heart, prostate and kidney disease by Mayo Clinic and other researchers. In a two year experiment, calcifying nanoparticles were cultured from the joint fluid of each participating patient, demonstrating that 100 percent of patients had the particles. The experiment was based on methods pioneered by Nanobac Scientists, Drs. Neva Ciftcioglu and Olavi Kajander. "After about 2 months of culture, nanoparticles appeared in the synovial fluids from all the patients to greater or lesser degrees," the study found. "These nanoparticles gradually increased in number and in size." Medical researchers have often theorized about the presence of a calcifying agent that generates associated painful inflammation in arthritis, but until now had never found one. Nanobacteria produce a calcium phosphate material known as calcification, which is shown in many studies, and cited in the Merck Manual of Diagnosis and Therapy, as provoking inflammation. "If self-proliferating nanoparticles exist in mammalian synovial fluids and membranes, then they may have an effect on many joint diseases," the study's authors concluded. "This is another example of independent researchers finding nanobacteria in patients with a specific disease condition," said Nanobac Co-Chairman Dr. Benedict Maniscalco, Fellow of the American College of Cardiology (FACC). "It shows the value of our research, which is the only work to explain how calcification occurs in diseases afflicting most of the aging population," Dr. Maniscalco added. The findings come on the heels of a paper published in the World Journal of Urology by Cleveland Clinic researcher Dr. Daniel Shoskes and Dr. Hadley M. Wood, which concluded that nanobacteria-like particles may play a ubiquitous role in prostate disease. Another independent paper published earlier this year in the journal Urology Research by Khullar et al reports induction of renal calcification by nanobacteria. Disclosure statement: Nanobac Pharmaceuticals did not fund the arthritis or Khullar studies, so the findings are independent. Link to study abstract at: http://www.nanobac.com/content/scientific-publications.htm About Nanobac Pharmaceuticals Nanobac Pharmaceuticals, Inc. is a life science company dedicated to the discovery and development of products and services to improve people's health through the detection and treatment of Calcifying Nanoparticles, otherwise known as "nanobacteria". The Company's pioneering research is establishing the pathogenic role of nanobacteria in soft tissue calcification, particularly in coronary artery, prostate, and vascular disease. Nanobac's drug discovery and development is focused on developing new and existing compounds that effectively inhibit, destroy or neutralize CNPs. Nanobac manufactures In Vitro Diagnostic (IVD) kits and reagents for the detection of Calcifying Nanoparticles. IVD products include the NANOCAPTURE(TM) and NANO-SERO(TM) ELISA assays and the Nano-Vision(TM) line of antibodies and reagents. Nanobac's BioAnalytical Services works with biopharmaceutical partners to develop and apply methods for avoiding, detecting, and inactivating or eliminating CNPs from raw materials. Nanobac Pharmaceuticals, Inc. is headquartered in Tampa, Florida. For more information, please visit our website at: http://www.nanobaclabs.com
The Center for Biofilms Engineering Both medical and industrial aspects of biofilms are discussed. Biofilms in prostate are not mentioned, but you can get a feel for what biofilms are and why they are so hard to combat. For anyone wanting to learn about biofilms go to: http://www.erc.montana.edu/CBEssentials-SW/bf-basics-99/bbasics-01.htm
Breaking the biofilm barrier: Will it help us conquer infectious disease? From Contemporary Urology, September 2001 Bacteria are ubiquitous in our environment. The vast majority of bacteria do not live in a free-swimming planktonic form, but rather in the self-produced, protective environment of a biofilm, which seems to explain why some infections are nearly impossible to eradicate. Spend a weekend unclogging bathroom pipes or slipping on the rocks in a mountain stream and you will understand some of the other ways biofilms affect us. The concept of biofilms is relatively new -William Costeiton coined the term in 1978. His early studies demonstrated the protective mechanisms of biofilms in providing an environment for bacteria away from white cells, antibacterials, and environmental stresses. His microscopic examination of biofilms identified bacterial colonies interwoven with a polysaccharide matrix -a previously unappreciated fact. Small channels in the biofilm matrix permit the flow of oxygen, nutrients, and waste necessary to maintain microbial activity and reproduction.
While we have effective antibiotics to kill planktonic bacteria, the reservoir of bacteria contained in biofilms does not respond well to current antibiotics because of the protective nature of these structures. The large clusters of bacteria that make up a biofilm are similar to chemotherapy-resistant malignant cells, in that organisms in the clusters' center are protected from antibacterial invasion by a "wall" of fellow bacteria. Additional research has pointed to biofilm as producing a slower bacterial metabolism, antibiotic-degrading enzymes, and even the ability to "pump" antibacterial agents out of the biofilm before they can have any effect on the bacteria. Indeed, bacteria in a biofilm environment can be up to 1,000 times more resistant to antibiotics than the same bacteria circulating in a planktonic state.* Uropathogens and other bacteria produce biofilms unique to their species. In the lung, for example, Pveudomonas aeruginosa use their flagella to attach to each other and to the organ surface, thereby initiating colony formation. Staphylococcus, a major biofilm-forming organism (see the photo), produces infections in wounds and on prosthetic devices such as penile prostheses and artificial urinary sphincters, and may also be involved in the initiation of catheter-borne infections. Each year, biofilm-related infections on catheters, prosthetic devices, urinary catheters and tubes, and contact lenses cost the medical industry billions of dollars. The Centers For Disease Control estimate that more than 65% of hospital-acquired infections have biofilms as an integral part of their morbidity and potential mortality. Prosthetic implant infections are an excellent ease in point. Even when antibiotic agents are used in high doses and are in direct contact with the prosthetic device, these difficult, high-morbidity infections are rarely eradicated, necessitating removal of the device and its accompanying bacteria-filled biofilm. The search for new antibiotic agents is admirable. However, the key to conquering complex urologic infections may lie instead in overcoming the bacteriaprotecting barrier effect of biofilm. Research on antibiofilm compounds is underway at a number of laboratories across the country, and in vitro models are now available to identify the structure, architecture, unique characteristics, and production of biofilm. Despite these efforts, solutions remain elusive. Perhaps the next generation of antimicrobials will include methods of targeting the specific genes and regulatory mechanisms in bacteria that allow them to produce biofilms. A combination of biofilm-emulsifying drugs and good antibiotics will help us take giant steps forward in our treatment of urologic infections. *McCarthy M. Breaking up the bacterial happy home. Lancet. 2001;357(9273):2032 Culley C. Carson, MD, Editor in Chief. Professor and Chief, Division of Urology, University of North Carolina at Chapel Hill, USA. CCC@medec.com
J. Curtis Nickel, MD, Kingston General Hospital, Queen's University, Kingston, Ontario, Canada; Robert J.C. McLean, PhD, Southwest Texas State University , San Marcos, Tex. [Infect Urol 11(6):169-175, 1998. © 1998 SCP Communications, Inc.]
Abstract Bacterial biofilms play an important role in the pathogenesis, persistence, and eventual treatment of urinary tract infections (UTIs). Bacterial biofilms are associated with catheter-associated UTIs, struvite calculogenesis, and chronic prostatitis, as well as other common UTI scenarios. The bacterial biofilm theory describes bacterial populations in natural and pathogenic ecological systems in terms of a floating or "planktonic" population of bacteria interacting with a more important matrix-enclosed "sessile" population of bacteria associated with or adherent to a surface. This theory helps explain some of the difficulties encountered in the diagnosis and treatment of many UTIs.
Introduction The relatively new theory of bacterial biofilm helps clinicians understand and perhaps explain some challenging areas in the pathogenesis, diagnosis, and treatment of urinary tract infections (UTIs). The urinary tract is a hostile environment for bacteria. Except for the distal urethra, it usually remains sterile. Most UTIs are caused by ascending colonization and/or infection by enteric bacteria of the perineum (in women, the vagina and introitus), the periurethral area, the urethra, the bladder, and, occasionally, the kidney. Infection results when the bacterial virulence factors overcome the numerous host defenses.[1] The biofilm mode of growth allows bacteria to exist in the urinary tract, resulting in many of the clinical infections.
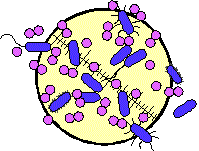
Bacterial Biofilm Direct microscopic observation has shown that many types of UTIs (eg, catheter-associated infections, struvite urolithiasis, continuous ambulatory peritoneal dialysis, and chronic prostatitis) are associated with biofilms (adherent populations of bacteria).[2,3] Biofilm formation occurs when microorganisms attach to a surface and, through growth and continuing colonization, spread over that surface. Bacterial attachment is facilitated by adhesins (structures on the bacterial cell surface, notably pili and extracellular polymeric substances [EPS]), whose primary function is adhesion.[4,5] As adherent cells grow, they form encapsulated microcolonies, which are small clumps of morphologically identical cells (often 2-10 cells) immediately adjacent to each other. Growth of adjacent microcolonies toward each other will lead to the development of a mature biofilm. This process and other aspects of biofilm biology are presented in greater detail elsewhere.[6]
"Sessile" bacteria within biofilms are physiologically quite distinct from unattached, "planktonic" bacteria. From a clinical perspective, the most significant features of biofilms are the resistance of the component microorganisms to antibiotics[7] and the immune response[8,9]; the creation of chemically distinct microenvironments (within which microorganisms can form calculi)[10]; and the potential for biofilm organisms to disseminate to other organs such as the kidney.[11] The biofilm mode of growth enables bacteria to persist in the urinary tract, but it is not unique to this system and occurs naturally in other environments.[12]
From a clinical perspective, antibiotic resistance is the most problematic and costly characteristic of biofilms. In vitro,[7,13] animal,[14] and clinical data[15] have confirmed that bacterial growth within thick biofilms adherent to urinary catheter material in a urine milieu confers a measure of antibiotic resistance on the sessile bacteria cells within the biofilm. Planktonic, or floating, cells in all these systems are completely eradicated at the antibiotic levels predicted by laboratory MIC studies. However, more than 100 times the MIC of antibiotics is required to eradicate cells within the bacterial biofilm.
Dispersion of the bacteria in the antibiotic-resistant biofilm further shows that the individual bacteria remain susceptible to low levels of antibiotics once they lose the protection of the biofilm itself. The increased resistance to antibiotic therapy in such biofilms may be secondary to poor antibiotic penetration into the biofilm matrix itself or decreased metabolic activity of bacteria deep within the biofilm, especially during periods of environmental (antimicrobial) threat.
Examination of biofilm structure (Fig. 1) provides a key to understanding certain features of biofilms. At first glance, a biofilm appears to consist of a copious, disordered mass of individual EPS-encased bacteria that adhere to a surface. However, investigations of biofilms by scanning confocal laser microscopy (SCLM) and other techniques[16] have shown their structure to be quite complex, often containing mixed populations of bacteria. Current research also suggests that cells within biofilms may actually communicate with each other, using cell-density signaling molecules, such as acyl homoserine lactone, for the purpose of coordinating metabolic activities and responses to stressors such as antibiotics.[17] Bacterial cells at the periphery of a biofilm, or those immediately adjacent to the water channels that separate them, are faster-growing than those within a microcolony because of greater access to nutrients. However, these faster-growing cells are more susceptible to attack by components of the immune system (such as lymphocytes) or by antibiotics than are the slower-growing cells buried within the microcolonies. Metabolic activity within a biofilm may also create a chemical microenvironment (eg, one conducive to struvite formation) that may be considerably different from that of the larger environment surrounding it.
Catheter-Associated Infections Bacterial biofilm has been associated with biomaterial-related sepsis such as that transmitted by urinary Foley catheters. Improved understanding of the pathogenesis of catheter-associated UTIs and the role of bacterial biofilms helps explain why catheterization is the most common cause of nosocomial infection in medical practice despite major technologic changes in catheter material, design, and collecting systems. In vitro,[18] animal,[19] and clinical studies[20] have shown that the pathogenesis of these infections involves an ascending, or creeping, bacterial biofilm. Observations from these experimental models have shown that bacteria form thick, coherent biofilms adherent to contaminated drainage spouts extending proximally into the drainage bag and subsequently into the catheter (Fig. 2). If a sterile, closed system is strictly maintained, the extraluminal route from the urethral meatus becomes the predominant route of catheter-associated bacteriuria. In the absence of antibiotics, it appeared that the ascending bacterial biofilm moved by 2 mechanisms: (1) rapidly dividing bacteria cells spreading along the catheter surface within the glycocalyx material of the biofilm and (2) planktonic or floating bacteria cells within the urine column leapfrogging just ahead of the adherent biofilm, perhaps assisted by the turbulence caused when the urine flow meets the biofilm front (saltatory movement).
The bacterial populations demonstrate a heterogeneity that is not evident from the culture results. Only a small proportion of the microorganisms including fungi, which are identified morphologically by scanning or transmission electron microscopy, are recovered by routine culture method. The nature of the bacterial biofilm adherent to the catheter can be appreciated by aspiration cultures of the planktonic bacteria being released from the biofilm. Usually, however, at this point in the process the bacteria are colonizing only the catheter surface and have not yet caused cystitis. As the adherent bacterial aggregate becomes larger, the now macroscopic bacterial biofilm can create flow problems by partially blocking catheter inlets and reducing the tubular diameter of the catheter lumen.
Planktonic bacteria are continually shed from the colonized catheter into the residual urine that is always present around the tip and balloon of the catheter. The final step from asymptomatic bacteriuria to symptomatic, catheter-associated cystitis involves actual adherence of these bacteria to the bladder surface. The indwelling Foley catheter appears to disrupt the bladder mucus or glycosaminoglycan layer by causing mechanical or chemical irritation and even erosion of the bladder mucosa, exposing surfaces that allow bacterial adherence. The synergistic activity between antibiotics and host defenses in these infections appears to clear the mucosal surfaces of adherent bacterial microcolonies much more easily than they are able to clear the very resistant bacterial biofilms adherent to the inert biomaterial of the catheter.[14]
Although the biofilm theory has helped explain difficult, Foley catheter-associated infection, currently the most effective means of reducing the incidence of these infections is to avoid indwelling, chronic catheterization, if at all possible, or at least to reduce the length of time the catheter remains in the bladder. In the future, new biomaterial research may allow production of materials that reduce bacterial biofilm attachment and enhance mucosal biocompatibility. New antibiotics are being developed that may be able to penetrate the bacterial biofilm.
Infection Stones of the Urinary Tract
Proteus mirabilis and other urease-producing bacteria are major causes of
UTIs.[21,22] The major risk in these UTIs is the development of urinary calculi,
which typically contain struvite (NH4MgPO4.6H20) and carbonate-apatite
(Ca10(PO4,CO3)6(OH,CO3)2) as the predominant mineral components. Infection
stones account for only 5% to 20% of all urinary calculi; however, they
represent a much more serious threat to the organs of the urinary tract than do
conventional metabolic stones because of their rapid growth and high rate of
recurrence. By blocking catheters and stents, infection stones cause both direct
renal damage as well as secondary effects.[23,24] Griffith and colleagues[25]
illustrated the fundamental role of urease in the pathogenesis of this
infection. Urea hydrolysis by bacterial urease activity elevates urine pH and
leads to Mg and Ca precipitation in the form of struvite and other minerals.
Ultrastructural examination of infection stones reveals the
It has been proposed that the bacterial biofilm mode of growth and its organic matrix (EPS) are largely responsible for initiating matrix deposition and crystal binding (within the urinary tract) and may be crucial in the process of crystal nucleation through creating an alkaline, metal (Ca and Mg)-saturated microenvironment.[27,28] Mature struvite stones have been likened to "fossilized biofilms"[29] within which the causative microorganisms are shielded from the effects of antimicrobial agents.[30] Of equal or greater importance, any dislodged or residual calculus fragments would contain viable organisms and could therefore act as seeds for the rapid development of new calculi. These 2 features explain the high recurrence of these calculi (about 50%) in spite of conventional surgical techniques.[21]
The knowledge of how struvite calculi are formed may encourage use of specific treatment measures, such as effective urease inhibition, and may also serve to emphasize the absolute necessity of removing all stone fragments and eradicating all organisms associated with the UTI. Otherwise the stones can and will recur.
Chronic Bacterial Prostatitis Although the diagnostic and classification systems in chronic prostatitis have been standardized, difficulties in differentiating chronic nonbacterial from bacterial prostatic inflammation are encountered in clinical practice. Additionally, the results of antibiotic treatment guided by culture and antibiotic-sensitivity data are dismal.[31,32] Current research on the association of bacterial biofilms associated with chronic prostatitis is unlocking some of the mystery surrounding this entity and will allow us to further rationalize diagnostic and therapeutic regimens.[33] It appears that bacteria in chronic bacterial prostatitis enter the prostate gland from the urethra (as ascending infection), perhaps assisted by turbulent urethral flow patterns and/or intraprostatic ductal reflux. Clinical studies involving a difficult group of patients with bacterial prostatitis who remain refractory to therapy have led to a further understanding of what is actually happening in the prostate gland during acute and chronic bacterial prostatitis.[33]
Once the bacteria enter the ducts and ascini of the prostate gland, they rapidly multiply, inducing a host response with infiltration of acute inflammatory cells into the ducts. In acute bacterial prostatitis, the entire prostate gland, or at least the major part of it, is involved in the inflammatory process. The ducts become engorged with infiltrate composed of dead and live bacteria as well as living and dying acute inflammatory cells, desquamated epithelial cells, and cellular debris. At this stage of the infection, because the majority of cells are planktonic, it is relatively easy to eradicate all the offending organisms with appropriate antibiotic therapy for complete resolution of the inflammatory process.
If bacteria persist from acute or, more likely, clinically subacute inflammation, they can form small, sporadic bacterial microcolonies or biofilms within the ductal system adherent to the epithelium (Fig. 4). These bacteria also produce an exopolysaccharide slime, or glycocalyx, that envelops these adherent microcolonies, and it appears that the microorganisms subsequently become very quiescent, undergoing a sort of "hibernation" when the environment becomes threatening. Surrounding these focal sites of bacterial persistence are areas of lymphocytic invasion with variable infiltration of plasma cells and macrophages. It appears that the persistence of bacteria in the prostate gland in these focal biofilms leads to persistent immunologic stimulation and subsequent chronic inflammation.
The traditional diagnostic routine -- although difficult, time-consuming, and expensive -- is the absolute key to diagnosis. It employs quantitative bacterial cultures of various urine segments and expressed prostatic secretion from the lower urinary tract.[31] However, urologists have abandoned this theoretically effective diagnostic culture technique because of a number of identifiable shortcomings.[32] The primary problem is that previous antibiotic therapy (prescribed prior to obtaining the proper specimens) appears to mask subsequent attempts at bacterial localization. Antibiotic therapy eradicates the planktonic bacteria within the ducts, and the very adherent bacteria in the biofilms do not appear to shed planktonic bacteria easily. This finding has been subsequently confirmed by biopsies of the prostate glands of prostatis patients with negative expressed prostatic secretion cultures when the organism is nonetheless detected within the prostate gland tissue culture.[33,34] Bacterial biofilms also appear to influence treatment results. The pharmacokinetics in noninflamed prostate glands have identified a number of antibiotics, including trimethoprim and the quinolones, as agents that can achieve reasonable levels within the prostatic fluid. Detailed pharmacokinetic studies of prostatitis in a rat model failed to show any significant difference in antibiotic levels in the inflamed prostatic duct.[35] It is much more likely that the adherent glycocalyx-encased, bacterial-biofilm mode of growth is conveying a relative resistance to the associated bacteria similar to that seen in catheter- and struvite-associated UTIs.
The knowledge that small adherent bacterial biofilms can exist deep within the prostate gland in chronic bacterial prostatitis should improve diagnostic and treatment regimens. It appears that an immunologic diagnosis -- based on the premise that although we cannot grow planktonic organisms, the sessile organisms continue to create an immune response that can be measured -- may improve differentiation of nonbacterial from bacterial prostatitis.[36,37]
Treatment regimens that deliver much higher antibiotic concentrations to the biofilm itself in the prostatic duct, theoretically improving treatment success rate (J.C. Nickel, unpublished date from ongoing investigation), are being developed. Finally, repetitive prostatic massage -- the historic treatment of prostatitis -- is making a comeback with clinicians.[38] Regular prostatic massage drains the obstructed ducts and perhaps converts more resistant sessile biofilm bacteria into sensitive planktonic forms that then become susceptible to treatment with newer, more potent antibiotics.
Biofilms Associated with Other UTIs Urethritis/Cystitis It is generally accepted that the route of infection and the development of cystitis begins with intestinal bacteria colonizing the introitus and the periurethral area with subsequent invasion of the bladder. An indigenous population of gram-positive bacteria, such as Lactobacillus species, exists within the introitus and vagina. They form a type of thin, living biofilm or bacterial barrier on the mucosa, which competitively inhibits the growth of pathogenic gram-negative bacteria.[1] However, if the protected bacterial population is defective, the invading pathogens may displace the favorable population and adhere to the introitus and periurethral area.
From colonization (as a thin biofilm on the periurethral and distal urethra) the bacteria instigate an ongoing battle against the intrinsic urinary tract defenses. When bacteria enter the bladder, individual floaters or planktonic bacteria are susceptible to the hostile environment of the urine, the mechanical forces of urine evacuation, and the bladder surface itself. If circumstances favor bacterial virulence over host defenses, uropathogens adhere to the mucosal surface in small, sporadic aggregates or microcolonies. These eventually may coalesce in patchy areas of bacterial biofilm adhering to superficial epithelial cells. At this point in the process, the resulting inflammation produces cystitis. Small doses of antibiotics prevent bacterial adherence and subsequent early biofilm formation and may be considered a prophylactic strategy.
Pyelonephritis When vesicoureteral reflux occurs, planktonic bacteria in the bladder secondary to asymptomatic bacteriuria or bacterial aggregates in cystitis can be flushed into the kidney. Alternatively, bacteria may ascend as a biofilm along the inner wall of a ureter paralyzed by the bacterial inflammatory process. Again, if bacterial virulence outweighs the host defenses of the kidney or if further defects such as renal scarring are present, the planktonic bacteria may adhere to the urothelium and papilla of the renal collecting system. Employing the P mirabilis model,[11] we have demonstrated that these bacteria may adhere in thin biofilms to the uroepithelium prior to invasion of the renal tissue and subsequent pyelonephritis.
Other Prosthesis-Related UTIs An indwelling ureteric stent can also develop a bacterial biofilm that can act as a continuing nidus for infection or that can build up to cause subsequent obstruction.[39] Penile prostheses used to treat impotence and external sphincter prostheses used to treat incontinence can also become secondarilyinfected. These infections are associated with bacterial biofilm attached to the biomaterial surface.[40] Similarly, infectious complications associated with devices used in nephrology (eg, peritoneal dialysis catheters, femoral and subclavian central venous catheters) are associated with bacterial biofilms.[41] These infections are not only resistant to antimicrobial therapy but can also cause life-threatening blockage of vital lines and tubes.
Summary Bacterial biofilms are implicated in the more serious and difficult UTIs encountered by clinicians in daily practice. Biofilm theory would attest to the futility of antibiotic therapy in these infections associated with inert prostheses (such as stents, catheters, and prostheses), and clinical experience has shown that the infections can eventually be successfully eradicated only by removal of the prostheses. Biofilms associated with obstructed prostate ducts in chronic prostatitis are problematic. The tenuous bacterial biofilms associated with cystitis and, perhaps, early pyelonephritis appear to be more easily eradicated by antibiotics compared with surface biofilms such as those associated with catheters because of the synergistic activity of antibiotics and host defenses against these bacterial biofilms. Understanding the bacterial biofilm concept and its association with host response and inert prostheses within the urinary tract is important with regard to improving diagnostic and therapeutic strategies in the management of these difficult UTIs.
En sammanställning av länkar som berör biofilms och nanobakterier: http://www.gak.co.jp/TIGG/TIGG44/44MR3e.html - See: ways to combat biofilm infections. http://helios.bto.ed.ac.uk/bto/microbes/biofilm.htm#crest http://www.novaflux.com/biofilm.html. http://instruct1.cit.cornell.edu/Courses/biomi290/Horror/Biof.tutorial.HTML http://www.cbsnews.com/stories/2003/01/13/tech/main536177.shtml http://abcnews.go.com/wire/Living/ap20030705_821.html http://www.mstp.northwestern.edu/m1jc_2002/ papers/Costerton_Hauser.pdf http://www.erc.montana.edu/CBEssentials-SW/director's%20message/default.htm http://www.courses.ahc.umn.edu/pharmacy/5825/uti.html%20 http://www.rnw.nl/science/html/bacteria010903.html http://link.springer-ny.com/link/service/journals/00345/bibs/9017006/90170345.htm http://drcranton.com/nanobacteria.htm http://www.pnas.org/cgi/content/full/97/21/11511 http://www.altcorp.com/AffinityLaboratory/biofilms.htm (and this one has 20 links for the research-minded) http://www.no-scalpelvasectomy.com/pdf/mens_letter_6.pdf (look here under: "medical intelligence") If a document is too wordy, trying searching the document ("control f" on your keyboard) for prostatitis, then prostate, then biofilm - 3 separate searches. The search will take you to the word you are searching for.
Nanobacterium sanguineum, Finland An unusual organism called Nanobacterium sanguineum has been identified in association with prostatic and other calcifications, although some controversy remains surrounding the characteristics and pathogenicity of this ultra-small and difficult to detect bacterium. It is unusual in many respects including its small size (about 1/1000 of the usual bacterial size) and its uniform association with layers of calcification, called a biofilm, with which it encapsulates and protects itself. It is believed by some to be associated with virtually all forms of calcification in the human body including prostatic calculi (stones), kidney stones, calcification of the ocular lens (cataracts) and arterial calcification (coronary and carotid artery disease). It has recently been shown to give a false positive test for Chlamydia. Very few labs are equipped to detect this organism and most physicians have still not heard of it. There is a commercial product being tested for treatment of coronary disease which uses a nightly rectal suppository of EDTA (this chelates calcium and unroofs the nanobacter making it vulnerable to the antibiotic) combined with oral enzymes and certain vitamins that augment the EDTA, and a bedtime dosage of tetracycline (doxycycline won't work) which kills the nanobacter once it is unroofed. The NanobacTx regimen is proprietary and only available through the group that developed it. You need to go to their website as I suggested and contact them to find out if any doctor in your area is able to write you the prescription for their program. Does it work for prostatitis? Anecdotally, the answer is yes. I tried it for myself since I have well documented prostatitis. I had severe, longstanding and intractable pain with ejaculation, and the urologists were unable to explain it or treat it effectively. It has made a huge difference for me. The 2 researchers from Finland who discovered the Nanobacterium sanguineum by serendipity and discovered its association with calcification were nominated for the Nobel prize in 2000 and are actively engaged in expanding the knowledge base. Ovanstående är utdrag från en artikel på en diskussionsgrupp av Dr. Bob Klein 2001-08-24, rklein42@aol.com Se även den kritiska artikeln http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1876495/?tool=pubmed
Prevalence of Human Anti-Nanobacteria Antibodies Suggest Possible Zoonosis, Sverige Martin Holmberg, MD, Dept. of Medical Sciences, Uppsala University, Uppsala/Sweden Nanobacteria are novel apatite mineral-forming agents found in human and animal blood and tissues, and arouse an antibody response. Earlier studies have shown that antibodies that react with nanobacteria also cross-react with bacteria from the related Bartonella group. To study possible cross-reactions further we tested serum samples from 400 Swedish healthy blood donors for nanobacterium and Bartonella antibodies. Antibodies to nanobaceria were assayed using an ELISA test supplied by Oy Nanobac Ltd (Kuopio, Finland). Bartonella IFA was performed using antigen preparations of Vero-cells co-cultivated with B henselae , B. henselae (”Marseille-strain”), B. elizabethae, B. quintana and B. grahamii. Four hundred serum samples were obtained from local blood banks at regional hospitals in Lund, Jönköping, Uppsala and Boden. The samples were collected during 1999 from healthy volunteers. Age and sex, as well as information on recent animal contacts were recorded in 393 cases. Of the 400 samples, 56 were positive (56/400 = 14%), using a cutoff value of 2 times the average of 4 negative controls. Seventeen of the samples were highly positive (17/400 = 4.25%) (> 3 times the average of negative controls). There was no statistically significant overlap between Bartonella- and nanobacterium-positive serum samples. Furthermore, there was no significant overrepresentation of gender, any age group or geographical locality in the donors who had nanobacterial antibodies. However, counting only the strongly positive serum samples, there was a significant overrepresentation in those donors that had recent contact with certain animals. The strongest association was seen in those with horse contact (p=0.007), but also in sheep (p=0.012) and cow contacts (p=0.026). In conclusion, we found no cross-reactions of serum samples positive to nanobacteria with five different Bartonella antigens. Our results thus indicate separate risk factors for exposure to Bartonella and nanobacteria. At this point they are largely unknown, but it is interesting to note that the most strongly seropositive cases to nanobacteria were associated with animal contacts.
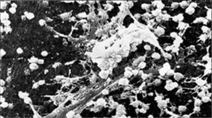
Kanske "elaka" nanobakterier i stark förstoring?
Tillbaka till Orsak till prostatit (på sidan Prostatasjukdomarna)
|